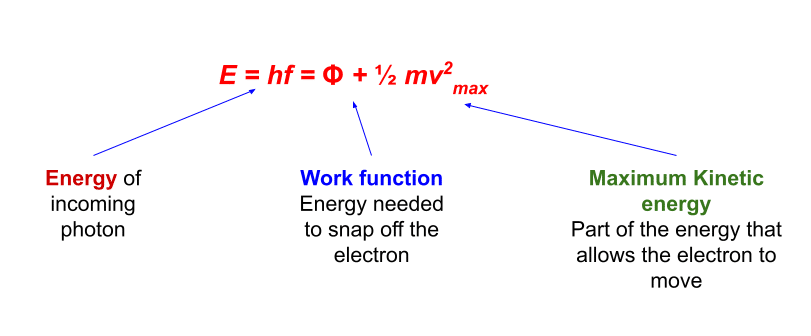
If you substitute 0 for m (because a photon is massless), and E hv (the. Einstein's E mc2 formula is actually a special case of the special relativity formula E2 p2c2 + m2c4, where p is momentum, m is rest mass, E is energy, and c is the speed of light. If the light has a high enough energy, the electrons will escape from the surface of the material. Although a photon is massless, it still has momentum. Different wavelengths (or colours) of light carry different amounts energy. When the light strikes the metal, its energy is transferred to the electrons. You can express this relationship in a formula by using the law. The photoelectric effect is all about kicking electrons out of metals, using light. a certain frequency or wavelength), the energy of the photons is given by Ehfhc, where h is Plancks constant. The applications of this effect are of course endless, as you can undoubtedly think of countless devices that involve detection of light. One interesting application is a device known as a photomultiplier tube. Suppose you wish to be able to detect and amplify very low intensities of light (in any part of the spectrum). As the light frequency increases, the kinetic energy of the electrons increases proportionally. This equation is read this way: "The electron charge multiplied by the stopping (electrostatic) potential is the potential energy change that barely stops the electrons with the greatest amount of kinetic energy, and this equals the energy given to the electron by the photon, minus the work function (the potential energy holding the electron to the surface of the conductor)." Where the gradient is the Plank constant (h) and the y intercept is the work function( f), the intercept on the x-axis is the threshold frequency f 0.=hf - \phi\] So plotting a graph of frequency (f) on the x-axis and maximum kinetic energy (E k) on the y-axis will give a straight line graph. Then if N N photons per second hit the cathode, the. For simplicity, lets assume every photon generates a photo-electron. Assuming there is a sufficient electric field, these electrons will make their way across to the anode, contributing current. The above equation can be rearranged into the from y=mx+c In the photoelectric effect, photons incident on the cathode cause the emission of electrons. Higher intensity means more photons per unit area. So we can see from the equation above that if the light does not have a big enough frequency (f) so that the photon has enough energy to overcome the work function ( f) then no photoelectrons will be emitted. The energy of each photon is Ehf, where h is Plancks constant and f is the frequency of the EM radiation. The energy of a photon of light = hf and the work function ( f)is the minimum energy required to remove an electron from the surface of the material. That electron is then shot out of the metal. When these particles collide, some of the energy of the photon is used to dislodge the electron. What happens is that the photon collides with the metal surface, hitting an electron. E k = the maximum kinetic energy of the emitted electrons in joules (J) A particle of light is called a photon, and when a photon has enough energy, it can actually knock an electron free.f = the frequency of the incident light in hertz (Hz). 
where E E is the energy of a photon of frequency f f and h h is Planck’s constant. 
h = the Plank constant 6.63 x 10 -34 J s In his explanation of the photoelectric effect, Einstein defined a quantized unit or quantum of EM energy, which we now call a photon, with an energy proportional to the frequency of EM radiation.The above two observation can only be explained if the electromagnetic waves are emitted in packets of energy (quanta) called photons, the photoelectric effect can only be explained by the particle behaviour of light. the maximum kinetic energy of the photoelectrons depended on the frequency of the light not the intensity of the light.the frequency of the light needed to reach a particular minimum value (depending on the metal) for photoelectrons to start escaping the metal The equation to calculate the energy of an emitted electron is: E hf -, where E is the energy of the electron emitted, h is the Plancks constant.The escaping electrons are called photoelectrons. 
The photoelectric effect occurs when light above a certain frequency (the threshold frequency) is shone on metals like zinc, this causes electrons to escape from the zinc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
